1 in 3
adults develop arrhythmias throughout their life, often without warning signs1
5X
more risk of stroke if you have AF1
20%
direct cost savings when implemented in routine care2
89%
of users feel instant peace of mind without the need for a GP appointment3

Seamless integration
We offer a range of integration options tailored to your needs

No additional hardware
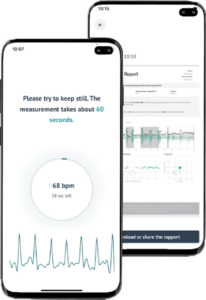
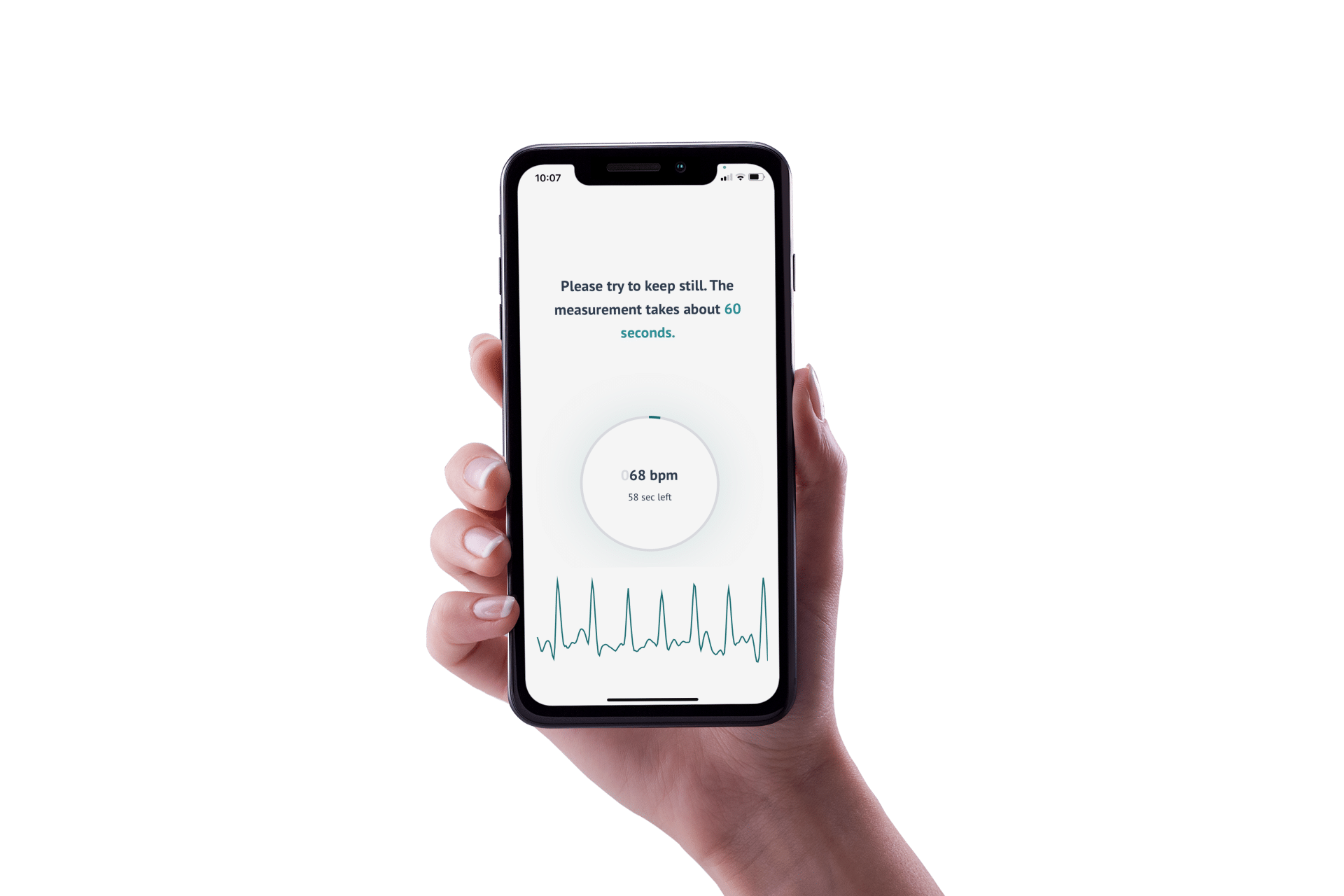
To perform measurements, users only need a smartphone

No regulatory hassle
Make use of our existing regulatory approvals and avoid regulatory delays
The possibilities of FibriCheck

FibriCheck partners with Pfizer and AlTibbi for the first of its kind atrial fibrillation awareness campaign in the UAE
FibriCheck is thrilled to announce its strategic partnership with Pfizer and AlTibbi, the leading digital health platform in the MENA region…

Belgian health insurer Helan offers FibriCheck for free to its clients
Since the beginning of 2022, several Belgian health insurers – including Helan – have started with a reimbursement of up…