Easy integration into any device with photoplethysmography (PPG) sensors opens up new possibilities for medical device and pharma companies
.

This increasing adoption of smartwatches and wearables (smart glasses, smart shirts, fitness trackers, etc.), along with growing numbers of people who want to be more involved in their own health care, has opened the door to new ways of managing a wide range of health conditions.
Detecting atrial fibrillation with wearables
Atrial fibrillation offers a compelling example. The most common arrhythmia to require treatment, atrial fibrillation (AF) affects a large part of the population. Approximately 9% of people over the age of 65, and about 2% of people under that age, have atrial fibrillation³ – but it can be very hard to diagnose. Episodes of AF can come and go, and some people with AF don’t experience any symptoms at all.
The problem is that untreated AF can lead to a stroke. In fact, 20% of all strokes can be directly attributed to atrial fibrillation. As the population ages, a greater number of people will develop AF.
Improving the diagnosis of AF, and subsequent treatment is key to patient and health system outcomes. The good news is that 75% of these types of strokes can be prevented by timely detection. Traditionally, AF was detected in a clinical setting, using expensive equipment. Now, new wearables with the ability to monitor heart rhythms and identify episodes of AF are in line to take over the torch.
The Apple Watch was the first in this field, but other wearables have joined the fight, with the innovative FibriCheck heart rhythm app taking the lead with more integration options and functionality.
FibriCheck heart rhythm app provides an extensive set of capabilities

The FibriCheck heart rhythm app offers a more extensive set of capabilities compared to other wearables options, providing users with deeper insights and a more comprehensive level of care. Key FibriCheck attributes include:
1. Closed, secure communications loop between patient and doctor
The FibriCheck app is available in two business models: a consumer model, where users review and manage their own heart rhythm data, sharing results with a doctor if and when they desire; and a physician model, where physicians prescribe or advise the FibriCheck app for their patients and actively monitor the heart rhythm data that is automatically shared with them. In both models, data is shared via a closed, secure communications loop, with information stored in a secured medical cloud.
2. PPG performance on wearable is equal to “gold standard” of single-lead electrocardiogram (ECG)
FibriCheck uses one robust algorithm to process data from both wearables and smartphones. In validation studies4, researchers found that the diagnostic performance of the FibriCheck app is equivalent to that of state-of-the-art single-lead ECG-based devices. FibriCheck has received FDA 510(k) clearance with the “substantial equivalence” determination (for this purpose deemed substantially equivalent to the AliveCor Heart Monitor.) The study5 that compared FibriCheck to the AliveCor Mobile ECG (which uses a single-lead ECG), found that FibriCheck had the same level of diagnostic accuracy. The results of a follow-up study for the wearable, directly comparing the performance of FibriCheck PPG with the Apple Watch ECG, will be presented by Lars Grieten, FibriCheck Co-founder and CEO, at the EHRA (European Heart Rhythm Association) 2020 Congress in Vienna, on March 30, as part of a session on technology and innovation. Visit the EHRA session to learn more, or sign up for our newsletter.
3. Semi-continuous heart rhythm monitoring (beta launch planned for end of March 2020)
In this mode, the FibriCheck app will check the user’s heart rhythm at various times during the day and night, performing multiple spot checks and providing reports on any heart rhythm abnormalities that are detected. It’s important to note that with other watches, including the Apple Watch ECG, this is a two-step process: the user receives a non-specific notification in the case of an irregular heart rhythm, suggesting that they manually take an ECG reading.
With the FibriCheck app this happens in a single automatic step: the spot check recording already contains all the data that a physician needs in order to identify a problem. Further, if a link has been established with the physician, the data is automatically shared. Given that many episodes of atrial fibrillation happen while people are sleeping, this extra level of insight is especially important in effectively diagnosing AF. Semi-continuous monitoring is not available on other wearable/smartwatch monitoring apps.
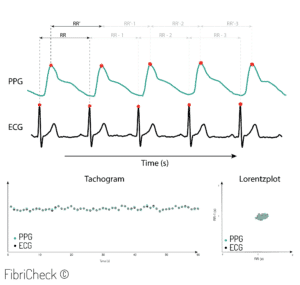
Comparing PPG to ECG
FibriCheck uses PPG (photoplethysmography) sensors to determine the heart rhythm of a user. Because most people are more accustomed to ECG technology being used to measure heart rate and to detect heart rate irregularities, there remains some unfamiliarity with the capabilities of PPG to detect heart rhythm disorders.
The Apple Watch, for example, uses both PPG and ECG technology to determine the presence of atrial fibrillation. First, the Apple Watch uses optical sensors to measure PPG signals. This method runs in the background, monitoring the user’s heart rhythm at specific intervals. When the Apple Watch app identifies a heart rhythm irregularity, it prompts the user with an “irregular rhythm” notification. On the Apple Watch, this irregular rhythm notification is generated by an algorithm that measures beat-to-beat variability. The heart rhythm is measured when the subject is sitting still, with a minimum interval of 15 minutes between 2 recordings. If 5 out of 6 recordings within a 48 hour period are positive, a notification is given to the user. The irregular pulse notification is logged in the mobile phone, but no additional (visual) information related to the recording is provided, meaning it is not possible to interpret these results or present them to a clinical practitioner6.
Once they receive a notification, the Apple Watch user is directed to conduct a second test by taking a single-lead ECG, which is activated by the user pressing their finger against the crown of the Apple Watch. After a connection is made, a 30 second ECG recording is carried out (spot-check) after which a result is shown, indicating either normal sinus rhythm, atrial fibrillation or unclassified.
The Apple Watch concept was validated in a large clinical study where a positive predictive value (PPV) of 84% was found. No results are known on the sensitivity. The Apple Watch ECG operates at a reported very high clinical accuracy (in the 95%-97% range)6.
Traditionally, the ECG signal is considered to contain the necessary information for a medical professional to make a diagnosis where the most commonly used PPG techniques are lacking these insights.
FibriCheck and PPG: offering more granular interpretation of results
However, FibriCheck analyzes the PPG results in a completely different, and much more comprehensive way. Because it uses more features than the beat-to-beat variation, it results in improved granularity compared to traditional PPG techniques. The FibriCheck algorithms not only enable the differentiation of heart rhythm disorders compared to single-lead ECGs, but also allow for the “reading” of the measurement result by medical experts for correct interpretation and analysis. For these reasons, the implementation of a FibriCheck PPG is closer to the working of a single lead ECG, compared to other PPG techniques.
Next steps with FibriCheck
The device-agnostic FibriCheck algorithm software can be installed on any wearable product, device or phone that uses PPG sensors, opening up new possibilities for wearables, medical device and pharmaceutical companies, looking for new ways to support patient empowerment and better heart health.
If you’d like to find out more about integrating the FibriCheck heart rhythm app into your product, application or practice, and would like to discuss the different integration approaches that you could take, contact us.
References
- eMarketer editors, “Older Americans Drive Growth of Wearables,” Dec. 13, 2018, retrieved from: https://www.emarketer.com/content/older-americans-drive-growth-of-wearables
- Deloitte editors, “2017 Global Mobile Consumer Survey: US edition, The dawn of the next era in mobile,” 2017, retrieved from: https://www2.deloitte.com/content/dam/Deloitte/us/Documents/technology-media-telecommunications/us-tmt-2017-global-mobile-consumer-executive-survey.pdf
- Centers for Disease Control and Prevention, Division for Heart Disease and Stroke Prevention, “Atrial Fibrillation Fact Sheet”.
- Proesmans T, Mortelmans C, Van Haelst R, Verbrugge F, Vandervoort P, Vaes B, “Mobile Phone–Based Use of the Photoplethysmography Technique to Detect Atrial Fibrillation in Primary Care: Diagnostic Accuracy Study of the FibriCheck App,” JMIR Mhealth Uhealth 2019;7(3):e12284, retrieved from: https://mhealth.jmir.org/2019/3/e12284/ DOI: 10.2196/12284 PMID: 30916656 PMCID: 6456825
- Vandenberk T, Stans J, Mortelmans C, Van Haelst R, Van Schelvergem G, Pelckmans C, Smeets CJ, Lanssens D, De Cannière H, Storms V, Thijs IM, Vaes B, Vandervoort PM, “Clinical Validation of Heart Rate Apps: Mixed-Methods Evaluation Study,” JMIR Mhealth Uhealth 2017;5(8):e129, retrieved from: https://mhealth.jmir.org/2017/8/e129/ DOI: 10.2196/mhealth.7254 PMID: 28842392 PMCID: 5591405
- https://www.apple.com/healthcare/docs/site/Apple_Watch_Arrhythmia_Detection.pdf
Created on February 19th, 2020 at 11:49 am
Last updated on January 6th, 2023 at 05:28 pm