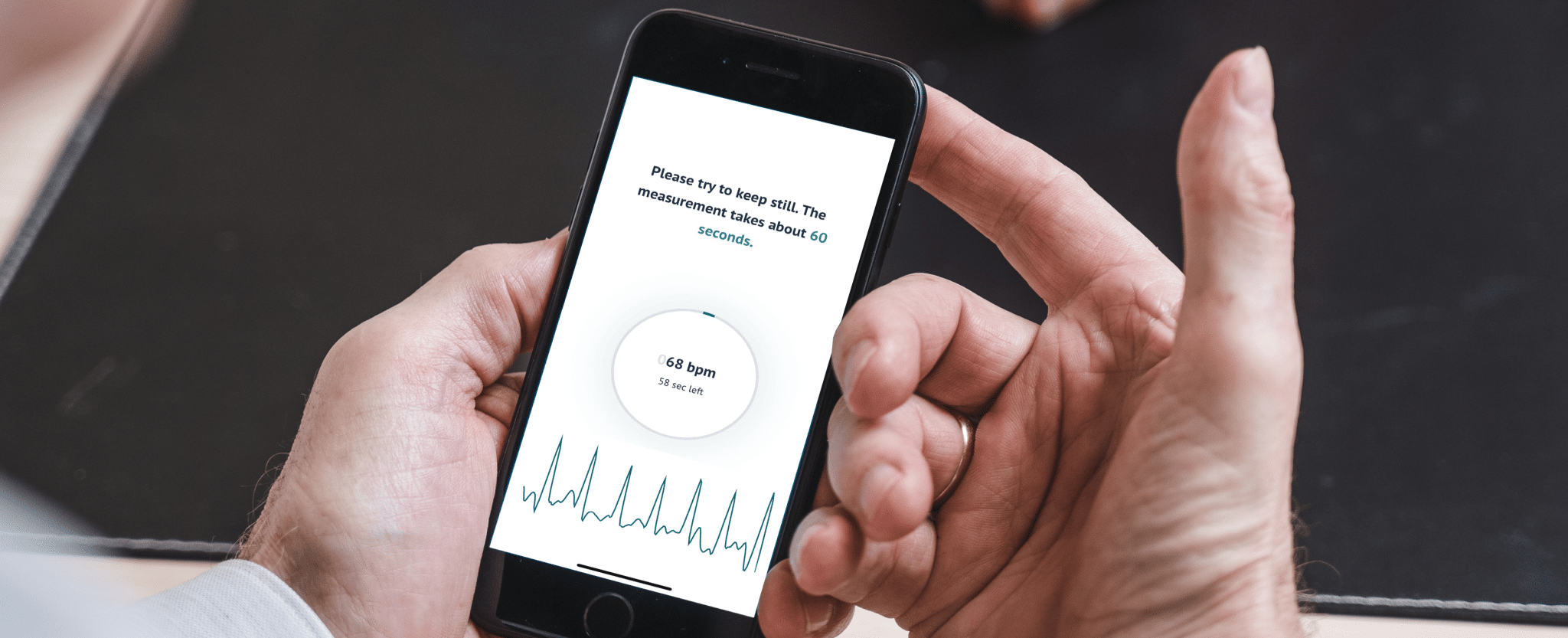
FibriCheck gains approval from Australia’s Therapeutic Goods Administration (TGA)
FibriCheck, the mobile app capable of detecting heart rhythm disorders, was granted an Australian Register of Therapeutic Goods Certificate by the Australian Government Department of Health last week. As the first stand-alone application of its kind to receive TGA clearance, this important milestone paves the way for FibriCheck, not only onto the Australian market but also the wider Asia-Pacific region.
Far from a formality
FibriCheck’s software had already been approved as a Class IIa medical device by the EU in 2017 and later by the US watchdog, the Food and Drug Administration (FDA) in 2018. And while these approvals made for a strong application to the TGA, obtaining clearance from the Australian authority was by no means a formality. The regulatory requirements for medical devices in Australia are some of the most stringent in the world and before any device is included in the country’s register of approved goods, it must undergo a lengthy and comprehensive process of audits and both pre- and post-market reviews.
A unique case
As far as medical devices for the detection of heart rhythm disorders are concerned, FibriCheck’s application to the TGA was a unique case. In fact, FibriCheck is the first and currently only TGA-approved software for heart rhythm disorders which uses a non-medical device such as a smartphone, smartwatch or other consumer wearable device equipped with a photoplethysmographic (PPG) sensor.
Using powerful algorithms developed over years of clinical research, FibriCheck measures user’s heart rhythms via a mobile or wearable device, alerting them to potential episodes of even difficult-to-detect disorders such as silent or intermittent atrial fibrillation. The data and reports generated can then be easily forwarded to the user’s physician for further analysis, diagnosis and treatment, if required. Read more about how FibriCheck works on our blog.
Backed up by data
Over the course of the application process, which began in May 2019, FibriCheck was able to compile extensive clinical data and documentary evidence demonstrating the app’s precision and reliability. Particularly in comparison with traditional methods of detecting heart rhythm disorders such as electrocardiograms (ECG), the data accrued over the past year makes an overwhelming case for the accuracy and above all convenience of the FibriCheck app.
Lars Grieten, CEO FibriCheck: “Over the past 12 months, we’ve been working hard on our roadmap to enable international market access for the FibriCheck product family. Receiving market clearance from the Australian Therapeutic Goods Administration (TGA) is an important first step towards marketing and distributing our products in the Asia-Pacific region. FibriCheck has had a strong regulatory focus from the beginning. And we have continued to build on those solid foundations ever since.”
A team effort
The application to the TGA was meticulously prepared by FibriCheck’s team of experts and closely followed up throughout the extensive 12-month review process.
Lars Grieten: “TGA approval would not have been possible without a coordinated team effort. It is a privilege to work with such a young and dynamic team whose professionalism is recognised with these major milestones.”
Needless to say, the entire team is excited by this latest achievement and looking forward to bringing FibriCheck Down Under… Stay tuned for more details.
Keen to learn more about FibriCheck?
Sign up for our newsletter and feel free to get in touch by email. We welcome your questions and feedback.
Created on June 8th, 2020 at 12:55 pm
Last updated on November 4th, 2020 at 09:21 am